Bearded Seal
Bearded Seal
Updated: July 2020
The bearded seal gets its name from the long white whiskers on its muzzle. Another English name for them, “square flipper”, refers to their large pectoral flippers which are flattened at the ends. These characteristics, plus the fact that the head appears small relative to the overall body size, make the bearded seal easy to distinguish from other northern seals.
Bearded seals are found throughout the Arctic and are the largest species of seal in the north being able to reach lengths of 2.1–2.7 m in length and weigh between 200–430 kg (DFO 2016). Males and females are difficult to distinguish, although in the spring the females tend to be slightly larger than the males. The largest bearded seal recorded was a female that weighed 432 kg (Cameron et al. 2010) and maximum body size is reached when the seal is 9–10 years old. Bearded seals may live for up to 30 years.
Abundance
Estimates suggest that there are approximately 500,000 to 1 million bearded seals across the Arctic, however the data available is rather poor.
Distribution
They are found throughout the Arctic and sub-Arctic, and are strongly associated with sea ice.
Relation to Humans
There is a subsistence hunt of bearded seals throughout their range, with some previous commercial hunt in Svalbard and Russia. There is also currently a small sport-hunt in Svalbard.
Conservation and Management
NAMMCO provides management advice in the member countries. Hunters must have a permit but there is no quota in both Greenland and Norway,
The most recent assessment (2016) listed the species as ‘Least Concern’ on the global IUCN Red List, as well as on the Norwegian national red list.

Bearded seal © Martha de Jong Lantink
Scientific name: Erignathus barbatus (Erxleben 1777)
Erignathus barbatus barbatus –“Atlantic” subspecies
Erignathus barbatus nauticus – “Pacific” subspecies
Faroese: Granarkópur
Greenlandic: Ussuk
Icelandic: Kampselur, Granselur, Kampur
Norwegian: Storkobbe
Danish: Remmesæl
English: Bearded seal, Square flipper
Lifespan
Bearded seals live to around 30 years old.
Average Size
They are typically 2.1-2.7m in length and 200-430 kg in weight.
Migration and Movements
Bearded seals stay around the ice edge, following it as it moves seasonally.
Feeding
They are benthic (bottom) feeders, eating mostly fish but also some invertebrates.
General characteristics

© Danita Delimont/Shutterstock.com
Bearded seal adults have dark grey or brown fur, often with reddish brown faces and pectoral flippers. The young are a lighter grey or brown with a pale face. Pups are born with a long lanugo coat (fine soft hair), which is grey to black in colour with lighter areas along their head and back, and sometimes rear flippers. This long coat is quickly shed during the nursing period.
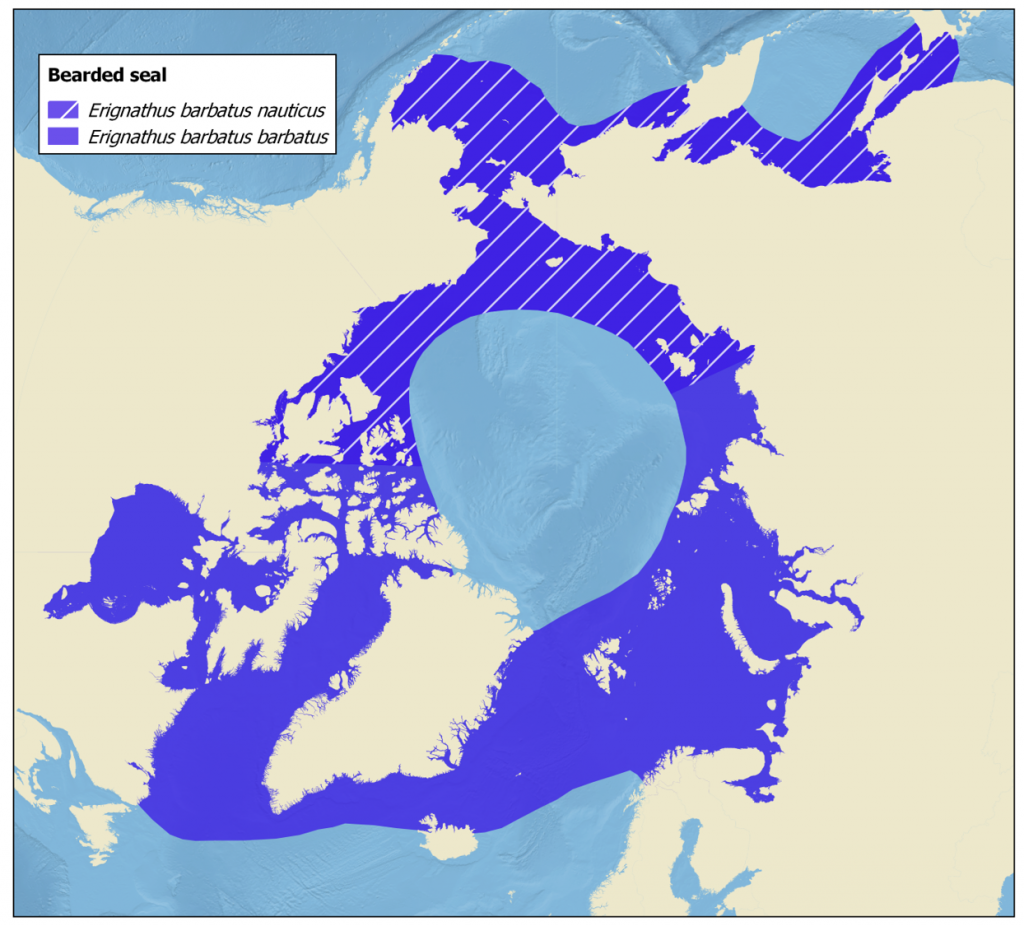
Researchers recognize two subspecies of bearded seal. The “Atlantic” subspecies (Erignathus barbatus barbatus) is found from the central Canadian Arctic eastward to the central Eurasian Arctic. On the other hand, the “Pacific” subspecies (E. barbatus nauticus) occurs from the Laptev Sea eastward, including the Sea of Okhotsk, to the central Canadian Arctic. The geographic distributions of these subspecies do not exhibit distinct gaps, and there is ongoing discussion regarding the precise boundary separating the two subspecies. There is support for recognizing two subspecies from genetics studies (Davis et al. 2008), as well as from studies of geographical differences in the calls of male bearded seals (Charrier et al. 2013, Risch et al. 2007).
Behaviour
Social behaviour
Bearded seals are not a social species and typically occur alone or in small groups throughout their range. During the breeding season, bearded seals exhibit certain social behaviors, with males engaging in producing various vocalizations to advertise their breeding condition (Mizuguchi et al., 2016) and/or defend their breeding territory (Charrier et al., 2013). Males are also aggressive toward other males during breeding, with both bubble-blowing displays and direct physical contact (Kovacs 2016).

© Ondrej Prosicky/Shutterstock.com
Habitat preference
Bearded seals maintain a close association with sea ice throughout their lives, particularly during critical life history periods concerning reproduction and moulting. They show a preference for areas of moving pack ice and open water, such as leads and polynyas, where the water depth is generally less than 200 meters. Nevertheless, researchers have observed bearded seals creating breathing holes in landfast ice and inhabiting deeper areas. In the open water season, these seals are entering river estuaries and hauling out on land (Stewart & Lockhart, 2005). Although their primary habitat is coastal regions, they can also be found in drifting pack ice situated far from the shore in shallow areas such as the Bering and Barents Seas (Kovacs et al., 2011).
Bearded seals overwinter near polynyas or other areas with leads and open water, or they stay near ice edges. The “North Water”, a large annual polynya between Canada and Greenland, is an important overwintering location for bearded seals. An aerial survey conducted over the eastern part of the North Water in April 2014 found an estimated 10,000 bearded seals there (NAMMCO 2015).
In some areas, as in the Bering and Chukchi seas, they undergo seasonal movements with the advance and retreat of sea ice (Kovacs 2016). In other parts of their range, they stay more or less in the same area year-round, making more local movements in response to ice conditions.
Sexual maturity and breeding
In general, bearded seals attain sexual maturity at 5‐6 years old for females and 6‐7 for males (Cameron et al. 2010).Surveys conducted in various Arctic locations have revealed that females, however, achieve sexual maturity between the ages of 3 to 7 years (Andersen et al., 1999). In Svalbard, females become mature at 5 years old (Kovacs 2016). Although males become mature at around the same age as females, they may not successfully breed until they are older.
The majority of mature females breed each year and produce one pup. Variation in pregnancy rates has however been recorded for various locations (Cameron et al. 2010). These variations may indicate differences in carrying capacity between the various habitats.
As in other phocid (earless) seals, the mother ovulates towards the end of the nursing period and mates after the she weaned her pup (Kovacs 2016). Similar to other seals, bearded seals undergo embryonic diapause with implantation occurring in late July or early August.

Two bearded seals on ice © Sergey Uryadnikov/Shutterstock.com
Pups
Pups are born on the ice, either on pack ice or on small floes of annual sea ice (Hindell et al. 2012). In Svalbard, bearded seals have also been seen to use loose pieces of glacier ice for birthing and nursing when sea ice is not available (Lydersen et al. 2014). Pupping occurs from late March to mid-May, with births occurring later in the year in the northern parts of the range. Peak pupping in Svalbard happens in early May, although newborn pups can occur there as late as August (Gjertz et al. 2000).
Bearded seal pups are larger than other species of seal, at about 130 cm long and 45 kg at birth (Richard 2001). Pups are born with long wavy lanugo hair, which is replaced by shorter, stiffer hair during the nursing period (Kovacs et al. 1996). The pup nurses for a period of 18–24 days, gaining weight very quickly at a rate of about 3.3kg/day (Lydersen et al. 1996). At weaning, its weight has often more than doubled to around 90–100 kg (Gjertz et al. 2000, Lydersen & Kovacs 1999).
Pup behaviour
Bearded seal pups are capable of swimming and following the adult female soon after birth (Hammill et al. 1994). A study of four nursing pups found that they spent about 53% of the time in the water and were submerged for 42% of that time (Lydersen et al. 1994). Six of the seven pups tagged in a study at Svalbard dived deeper than 448 m by the time they were 2 months old (Gjertz et al. 2000). This early swimming ability may have evolved so that the young can escape predation by polar bears, the bearded seal’s main predator. There is also evidence that pups are learning to catch and feed on small prey while they are still nursing (Lydersen et al. 1996).
Nursing females spend over 80% of their time in the water while caring for a dependent pup and only haul-out to nurse three times a day on average (Lydersen & Kovacs 1999). This might be a response to polar bear predation, as that a pup alone on the ice is less conspicuous than both a pup and mother.
Vocalisations

Bearded seal vocalising © Sergey Uryadnikov/Shutterstock.com
Adult male bearded seals produce very audible and musical vocalisations. In the Beaufort Sea, they were heard to produce calls year round, with activity increasing with the formation of pack ice in the winter and peaking in the spring, coinciding with the mating season (MacIntyre et al. 2013). These vocalisations (the most predominant termed “trills”) have been estimated to be detectible from as far as 25–45 km away (Jones et al. 2014). Female bearded seals also vocalise, although not to the same extent (Mizuguchi et al. 2016).
Individual males have distinct vocalisations, which could be both a means of defending their territory (Charrier et al. 2013) and of advertising their breeding condition (Mizuguchi et al. 2016). The duration of their vocalisations has been seen to differ depending on their mating strategy.
Male bearded seals return to the same breeding location year after year (Jones et al. 2014). Some males occupy and defend the same territory over a series of consecutive years, while other males exhibit a “roaming” pattern, returning to the same general area but not to specific display locations (Van Parijs & Clark 2006, Van Parijs et al. 2004). These roaming males may be young males or mature males in poorer condition that are unable to maintain a territory (Van Parijs & Clark 2006).
The vocalisations of each type of strategy varied for different locations – in Alaska, the roaming males had the longest calls, while in Svalbard, the territorial males did (Van Parijs & Clark 2006).
Males are aggressive towards each other during the breeding season, with displays of bubble-blowing and physical combat, indicating that the mating system is likely polygynous (Kovacs 2016). Mating occurs in the water, making it difficult to observe behaviour.
Moulting usually takes place after mating, between March and May, depending on the location, although it can occur anytime between April and August (Kovacs 2016). While moulting, they spend much of their time hauled out (Gjertz et al. 2000).
Food and Feeding
Bearded seals are primarily benthic (bottom) feeders, but their diet varies by age, location and season. They have been characterised as a “foraging generalist” able to prey on a wide variety of items throughout their circumpolar range (Antonelis et al. 1994). See Cameron et al.(2010) for a full list of prey items.
Major food items
Fish constitute a major portion of the diet. In 15 of 19 bearded seals from the Canadian high Arctic, whose stomachs contained more than 1 kg food, fish made up over 90% of the wet weight (Finlay & Evans 1983). Twelve different species of fish were present in those stomachs, with sculpins (Cottidae) and arctic cod (Boreogadus saida) the most common. A study of the stomach contents of 78 bearded seals in the Bering Sea found that 86% of the stomachs contained fish, including capelin (82%) and gadoids (64%) (Antonelis et al. 1994).

Capelin
Other benthic invertebrates are important in the diet. Bearded seals can capture prey on the bottom or use their broad muscular snouts to extract prey from the substrate by powerful suction and water-jetting (Marshall 2016). Their long mystacial vibrissae (whiskers) aid them in locating and identifying buried prey. The main invertebrate prey taken are benthic polychaetes, crustaceans and molluscs. In Svalbard, benthic gastropods and decapods make up from 50–80% of the bearded seal diet (Hindell et al. 2012).
The whelk Buccinum and the shrimp Sclerocrangon boreas accounted for most of the invertebrate component of the diet in the Canadian High Arctic. Clams, cephalopods, anemones, sea cucumbers, polychaete worms and other invertebrates occurred in small amounts (Finlay & Evans 1983).
Variations
Bearded seal diets have demonstrated to change with changing ice conditions. In Svalbard, seal diets consisted of more pelagic fish species and fewer benthic invertebrates in the years with the most extensive fast ice, while the opposite was seen in the years when the fjords were relatively ice free (Hindell et al. 2012).
In the central Bering Sea, Antonelis et al. (1994) detected no differences between the diets of males and females or between adults and juveniles, indicating no apparent change in the diet or feeding habits according to sex or age.
Predators
Polar bears are the main predators of bearded seals, although walruses, killer whales and Greenland sharks occasionally hunt them as well (Kovacs 2016, Cameron et al. 2010, Cleator 1996). Walruses appear to primarily take young seals, although it is not clear if they are preying directly on them or eating them as carrion (Cleator 1996).

Adult polar bear (Ursus maritimus) feeding on the remains of a bearded seal kill, Svalbard Archipelago, Norwegian Arctic © Nora Yusuf/Shutterstock.com
Health and parasites
Throughout their range, a wide variety of parasites were found in bearded seals. Parasitic worms are prevalent, including various species of cestodes, trematodes, nematodes and acanthocephalans. For a complete species list you can consult Cameron et al. (2010). The organs most commonly infected by helminth species are the stomach and intestinal tract, as well as the heart, gall bladder and lungs. Larvae of the nematode Trichinella have also been found in bearded seals (Forbes 2000). This parasite can infect humans through the consumption of raw or undercooked meat and cause trichinosis.
Protozoan parasites can also occur in some bearded seals. Intestinal contents from four bearded seals collected from Nunavik, Quebec, were analyzed for the presence of Giardia duodenalis and Cryptosporidium spp. Giardia duodenalis was present in three of the four seals, but Cryptosporidium spp. were not found (Dixon et al. 2008).
Relatively little is known about viral or bacterial diseases in bearded seals, or to what extent these might affect seal mortality.
Stock distribution, habitat and migrations
Bearded seals have a circumpolar distribution throughout much of the Arctic and sub-Arctic, and generally use habitats south of 85º N. Their distribution is influenced by both their strong association with sea ice, which they need for critical life history activities such as reproduction and moulting, and by their need for areas of high benthic productivity for feeding. They typically occur alone or in small groups, in a patchy, low density distribution. Distinct stocks are not currently recognised.
In some areas (e.g. the Bering and Chukchi seas), they undertake seasonal movements with the advance and retreat of sea ice (Kovacs 2016). In other parts of their range, they stay more or less in the same area year-round.
Bearded seals exhibit a preference for areas characterized by moving pack ice and open water, typically with water depths below approximately 200 meters. However, they have also been seen to maintain breathing holes in landfast ice or occupying deeper areas. In the open water season, bearded seals have been seen to enter river estuaries and haul out on land (Stewart & Lockhart 2005).
The North Atlantic
The Atlantic subspecies of bearded seals inhabits in Hudson Bay and much of the eastern Canadian Archipelago south to southern Labrador, as well as along both coasts of Greenland. They also occur along the north shore of Iceland, within the Svalbard Archipelago, and in the Barents Sea and across much of the north coast of the Russian Federation east to the Laptev Sea (Kovacs 2016). Individual seals have occured as far south as Portugal on the European side of the Atlantic. On the North American side, a bearded seal was seen in the harbour at Gloucester, Massachusetts (Sardi & Merigo 2006). Although bearded seals are not native to Iceland, they do occasionally show up, typically along the north or northwest coast (Hauksson & Bogason 1995).
North Atlantic Stocks
Biologists use the term stock to describe a subpopulation of a species that is largely reproductively isolated from others of the same species. As a result of this reproductive isolation, stocks can usually be differentiated genetically if they have been isolated for a sufficient length of time. Other means of stock differentiation include morphometrics (body size and shape), concentrations of pollutants or rare elements in tissues, behaviour (including vocal dialects), and patterns of seasonal movement. A key feature of this as a management concept is that the hunting and possible depletion of one stock should have little or no effect on a neighbouring stock.

Note the square front flippers characteristic of bearded seals© Smudge 9000
Bearded seals occur throughout the Arctic in a patchy, low density distribution. There may be distinct stocks or breeding populations, however, not enough is known at this time in order to be able to delineate them and none are currently recognised.
Two subspecies of bearded seal are recognised: Erignathus barbatus barbatus and Erignathus barbatus nauticus. The “Atlantic” subspecies–E. b. barbatus– inhabits the central Canadian Arctic east to the central Eurasian Arctic while the “Pacific” subspecies–E. b. nauticus–occurs from the Laptev Sea east (including the Sea of Okhotsk) to the central Canadian Arctic. There is still some discussion about where the boundary between the two subspecies should lie, however, there is support for recognising two subspecies from genetics studies (Davis et al. 2008), as well as from studies of geographical differences in the calls of bearded seals (Charrier et al. 2013, Risch et al. 2007).
Current abundance and trends
Current population numbers and trends for bearded seals are not well known. There are several factors that make it difficult to accurately assess bearded seal populations. Firstly, their remote and broad distribution makes surveying them expensive and logistically challenging. As a result, population surveys for these and some other Arctic marine mammals are “infrequent, incomplete or simply non-existent” (Kovacs et al. 2011). Furthermore, the range of the species spans several political boundaries and since there has not been much international cooperation to conduct range‐wide surveys, estimating current abundance is difficult.
Estimates of the total world population of bearded seals have ranged from 500,000 to around 1 million (Cameron et al. 2010). Laidre et al. (2015) summarised known population data for bearded seals throughout their range, but described the data available as “poor and outdated”. The methods used to arrive at bearded seal population numbers have varied with time and location, making comparison of the numbers difficult. Because of this, there are no reliable quantitative estimates of population trends for the species.
For most of the range of the Atlantic subspecies (E.b. barbatus) no population numbers or trends are known (Laidre et al. 2015). Cleator (1996) suggested a minimum population of 190,000 bearded seals for Canadian waters, but this number was based on summing different indices made between 1958 and 1979, rather than any direct population survey. The Greenland Institute of Natural Resources estimates that there are approximately 250,000 of the E.b. barbatus subspecies (GINR 2016).
Stock status
Bearded seals occur throughout the Arctic in a patchy, low density distribution. There may be distinct stocks or breeding populations, however, not enough is known at this time in order to be able to delineate them and therefore none are currently recognised.
There is subsistence hunt of bearded seals throughout their range, although this is at low levels relative to other seal species. Catch rates for bearded seals in all countries are considered small relative to the local populations and hunting is believed to have little impact on abundance (Cameron et al.2010). There is a general belief that the bearded seals’ distribution at relatively low densities across a vast area contributes to their protection from overexploitation (GINR, 2016).

© NOAA
Worldwide, the status of bearded seals in the IUCN Red List category is “Least Concern” (Kovacs 2016). On Greenland’s Red List, categorises the bearded seal as “Not Threatened” (GINR 2016), while in Canada the Committee on the Status of Endangered Wildlife in Canada (COSEWIC) defines the status as “Data Deficient” (DFO 2016).
In 2012, bearded Seals were listed under the US Endangered Species Act following a detailed review of their status (Cameron et al. 2010). The listing was not due to current population concerns, but because of the threats posed from climate change and increasing industrial activities in the Arctic.
Management
There is no international governing body that regulates the hunting of bearded seals. Advice on the management of bearded seals can be provided by the North Atlantic Marine Mammal Commission (NAMMCO). If requested by a member country, the NAMMCO Scientific Committee will prepare advice on the size of a sustainable hunt based on existing data, which the Management Committee will then consider when it proposes measures for conservation and management to member countries.
Greenland
In Greenland, Government of Greenland regulates the management of marine mammals and local municipalities carry it out by putting in place by-laws restricting hunting by area, season, or hunting method. All hunters must have a permit and provide catch records.
Bearded seals may be hunted year-round and there are no Total Allowable Catches (TACs) or allocations set. There are some restrictions on the hunting of seals in Melville Bay Nature Reserve and in the national park in North and East Greenland (Cameron et al. 2010).
Management advice is provided to the Greenland Government through NAMMCO.
Norway
Bearded seals don’t usually occur along the mainland coast and the Norwegian Marine Resources Act sets no quotas for the hunting of this species. The Svalbard Environmental Protection Act manages the hunt of bearded seals in Svalbard waters (Cameron et al. 2010).
Licensed hunters can shoot bearded seals in Svalbard during the open season, outside of protected areas such as nature reserves and national parks. There is no TAC for Svalbard and only a small number of seals are taken each year.
Canada
The federal Department of Fisheries and Oceans and regional resource management boards, such as the Nunavut Wildlife Management Board, co-manage bearded seals in Canada. The 2011-2015 Integrated Fisheries Management Plan for Atlantic Seals (DFO 2016) includes provisions for bearded seals, although no Total Allowable Catches (TACs) or allocations have been set at present. If there was to be any commercial catch, licences and permits would be required. However, the catch of bearded seals for subsistence purposes is permitted year-round (DFO 2016).
Russia
In Russia, the “Law of Fisheries and Preservation of Aquatic Resources” provides for subsistence hunt of seals by aboriginal Russian peoples and a TAC for the commercial catch is determined annually by the Russian Federal Fisheries Agency. Russia is the only nation to set a quota on the number of bearded seals that can be hunted (Kovacs 2016).
Recent Catches
There is currently a legal subsistence catch of bearded seals throughout their range (Laidre et al. 2015). Bearded seals have a tough, flexible hide which is valued for many purposes, such as lines or rope, kayak coverings, and kamik (boot) soles while the meat is also suitable for human and dog food (Hovelsrud et al. 2008).
Greenland
In Greenland, hunting of bearded seals takes place throughout the year. During the winter in North Greenland they are hunted by their breathing holes in the ice and by netting close to shore. In spring they are hunted while basking on the sea ice and in summer they are hunted in open water from boats (Hovelsrud et al. 2008).
From 2005 to 2007, between 1454 and 1773 bearded seals were taken in Greenland (NAMMCO 2009). More recently (2011 to 2013), catches have dropped slightly, with between 990 and 610 taken in West Greenland and 312 to 186 in East Greenland (NAMMCO 2014).
These figures do not include seals that were “struck and lost”, or not retrieved during the hunt.
Norway
Bearded seals were hunted commercially around Svalbard for several centuries, although they were taken more opportunistically in the hunt for other species. Currently, licensed hunters can legally catch bearded seals in Svalbard outside of the breeding season, and they take a few seals each year. Hunting of bearded seals is prohibited in all of Svalbard’s national parks or nature reserves.
Iceland
Bearded seals occasionally appear near Iceland, where local hunters opportunistically hunted by local hunters (Hauksson & Bogason 1995).
Canada
In Canada, bearded seals are hunted year-round for subsistence purposes. This occurs from the ice in winter and from boats during the open water season. The Nunavut Wildlife Harvest Study, which ran from June 1996 to May 2001, documented hunts of from 584 to 786 bearded seals a year in Nunavut. The five year average was 735 seals (Priest & Usher 2004).
Russia
Bearded seals have been commercially hunted by Russia in the Sea of Okhotsk and the Bering Sea, with annual catches exceeding 10,000 animals in some years during the 1950s and 1960s. There were indications of possible overexploitation in these areas , which led to a suspension of ship‐based sealing in both the Sea of Okhotsk and the Bering Sea from 1970 to 1975, and the imposition of annual total allowable catches (TACs) (Cameron et al. 2010). In 2010, the TAC for the Sea of Okhotsk was 2,100, while TACs for the White and Barents Seas in 2010 were 20 and 50 respectively, and 150 for the Kara Sea. (Cameron et al. 2010). None of the TACs include hunting for subsistence purposes.
Catches in NAMMCO member countries since 1992
| Country | Species (common name) | Species (scientific name) | Year or Season | Area or Stock | Catch Total | Quota (if applicable) |
|---|---|---|---|---|---|---|
| Greenland | Bearded seal | Erignathus barbatus | 2023 | West Greenland (Norhwater, Baffin Bay, Davis Strait) | 590 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2023 | East and South Greenland | 94 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2023 | Total | 684 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2022 | West Greenland (Norhwater, Baffin Bay, Davis Strait) | 891 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2022 | South and East Greenland | 33 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2022 | Total | 924 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2021 | West Greenland (Northwater, Baffin Bay, Davis Strait) | 690 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2021 | South and East Greenland | 136 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2021 | Total | 826 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2020 | South and East Greenland | 116 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2020 | West Greenland (Northwater, Baffin Bay, Davis Strait) | 878 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2020 | Total | 994 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2019 | South and East Greenland | 872 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2019 | West Greenland (Northwater, Baffin Bay, Davis Strait) | 161 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2019 | Total | 1033 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2018 | East | 223 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2018 | West | 1049 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2018 | Total | 1272 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2017 | East | 150 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2017 | West | 991 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2017 | Total | 1141 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2016 | East | 244 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2016 | West | 1091 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2016 | Total | 1335 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2015 | East | 274 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2015 | West | 1081 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2015 | Total | 1355 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2014 | East | 214 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2014 | West | 1145 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2014 | Total | 1359 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2013 | East | 213 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2013 | West | 903 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2013 | Total | 1116 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2012 | East | 164 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2012 | West | 936 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2012 | Total | 1100 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2011 | East | 200 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2011 | West | 1102 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2011 | Total | 1302 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2010 | Total | 1403 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2009 | Total | 1258 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2008 | Total | 1437 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2007 | Total | 1566 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2006 | Total | 1773 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2005 | Total | 1454 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2004 | Total | 1362 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2003 | Total | 1708 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2002 | Total | 1394 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2001 | Total | 2205 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 2000 | Total | 2695 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1999 | Total | 2336 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1998 | Total | 2354 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1997 | Total | 2349 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1996 | Total | 2123 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1995 | Total | 1900 | No quota |
| Greenland | Bearded seal | Erignathus barbatus | 1994 | Total | 1927 | No quota |
| Iceland | Bearded seal | Erignathus barbatus | 2023 | Iceland | 0 | |
| Iceland | Bearded seal | Erignathus barbatus | 2021 | Iceland | 0 | |
| Iceland | Bearded seal | Erignathus barbatus | 2020 | Iceland | 0 | |
| Iceland | Bearded seal | Erignathus barbatus | 2019 | Iceland | 0 | |
| Iceland | Bearded seal | Erignathus barbatus | 2018 | Iceland | 0 | |
| Iceland | Bearded seal | Erignathus barbatus | 2011-2017 | Iceland | N/A | |
| Iceland | Bearded seal | Erignathus barbatus | 2010 | Iceland | 1 | |
| Iceland | Bearded seal | Erignathus barbatus | 2009 | Iceland | N/A | |
| Iceland | Bearded seal | Erignathus barbatus | 2003 | Iceland | 2 | |
| Iceland | Bearded seal | Erignathus barbatus | 2002 | Iceland | 4 | |
| Iceland | Bearded seal | Erignathus barbatus | 1992-2001 | Iceland | N/A | |
| Norway | Bearded seal | Erignathus barbatus | 2023 | Svalbard | 12 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2022 | Svalbard | 27 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2021 | Svalbard | 30 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2020 | Svalbard | 15 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2019 | Svalbard | 11 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2018 | Svalbard | 18 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2017 | Svalbard | 23 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2016 | Svalbard | 34 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2015 | Svalbard | 17 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2014 | Svalbard | 21 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2013 | Svalbard | 33 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2012 | Svalbard | 33 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2011 | Svalbard | 12 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2010 | Svalbard | 12 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2009 | Svalbard | 11 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2008 | Svalbard | 21 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2007 | Svalbard | 23 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2006 | Svalbard | 19 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2005 | Svalbard | 30 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2004 | Svalbard | 30 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 2003 | Svalbard | 2 | No quota |
| Norway | Bearded seal | Erignathus barbatus | 1992-2002 | Svalbard | N/A | No quota |
This database of reported catches is searchable, meaning you can filter the information, e.g. by country, species or area. It is also possible to sort it by the different columns, in ascending or descending order, by clicking the column you want to sort by and the associated arrows for the order. By default, 10 entries are shown, but this can be changed in the drop-down menu, where you can decide to show up to 100 entries per page.
Carry-over from previous years are included in the quota numbers, where applicable.
You can find the full catch database with all species here.
For any questions regarding the catch database, please contact the Secretariat at nammco-sec@nammco.no.
EU sealskin ban and the Inuit exemption
In 1983, in response to concerns about the Canadian commercial harp seal hunt, the European Economic Community imposed an import ban on all harp seal whitecoat products. The ban, which was initially for 2 years, has been extended since then and was expanded in 2009 to include all seal products (DFO 2016).
Exceptions to the ban include products resulting from Inuit/indigenous hunts; products for the sole purpose of sustainable management of marine resources; and products for the personal use of travelers to the EU. As well, the ban does not apply to seal products trans-shipped through the EU to non-EU destinations (DFO 2016).
In order for seal products to be exempted from the ban and placed on the market in the EU the Inuit or indigenous hunt must meet certain conditions. The hunt must:
- be traditionally conducted by the community
- contribute to the community’s subsistence in order to provide food and income and not be primarily conducted for commercial reasons
- pay due care to animal welfare, while taking account of the community’s way of life and the subsistence purpose of the hunt (EUR-Lex 2016).
By-catch
By-catch, or entanglement in fishing nets or other gear, is not a significant source of mortality for bearded seals as these seals generally do not occur in areas where commercial fishing is taking place. Iceland has reported a total of 14 by-caught bearded seals between 2002 and 2018 in lumpsucker nets and gillnets (National Progress Reports Iceland). From 2002 to 2006, commercial fisheries in the Bering Sea – Aleutian Islands recorded an average annual mortality rate of one bearded seal(Cameron et al. 2010).
Struck and lost
Struck and lost refers to seals that are struck and assumed to be killed during the hunt, but are not recovered. These rates vary greatly depending on the age of the seal taken, the hunter’s experience, and the weather conditions at the time of the hunt. Cameron et al. (2010) suggests struck and lost rates in the Alaskan hunt are between 25–50%. This is likely based on Cleator’s (1996) estimates that struck and lost may be up to 50% in open water and approximately 25 % on ice.
Climate change
Bearded seals are very dependent on sea ice year-round, requiring stable ice pans in late spring for raising pups and moulting, as well as summer ice for resting. Ice for the pupping and nursing periods must also be in suitable areas, i.e. over shallow waters with a rich benthic community to provide food at this important stage of the life cycle (Kovacs et al. 2011).

© Peter Prokosch/GRID Arendal
Global climate warming is currently causing major reductions in both the extent and duration of sea ice cover in the Arctic, which poses a threat to bearded seals and other marine mammals that depend on ice. Bearded seals will lose critical pupping and nursing habitat if suitable ice floes are not available (Hindell et al. 2012, Kovacs et al. 2011), which could result in a lowering of the carrying capacity of their habitat (Laidre et al. 2015).
Bearded seals may also have to move further north in order to find suitable ice, restricting their distribution and range. This could result in increased competition for food, which may intensify if other temperate species also move north due to habitat/climate changes (Kovacs et al. 2011).
Around Svalbard, bearded seals have been observed using pieces of calved glacier ice as haul-out platforms, possibly in response to a loss of sea ice (Lydersen et al. 2014). In other areas, bearded seals use land as a summer haul- out platform, and in these areas they may be better able to cope with an increasing loss of sea ice than other Arctic seals. However, the current lack of baseline data for bearded seals makes monitoring any current or future changes in their populations due to climate change difficult.
Shipping / Oil spills
Increased ship traffic poses another potential threat to bearded seals due to reduced ice cover and more open water as a consequence of climate change. The dramatic decline in Arctic sea ice over the past few years has raised the possibility of regular cargo traffic using both the Northern Sea Route and the Northwest Passage (Laidre et al. 2015, Hovelsrud et al. 2008). Resource extraction and tourism activities will likely increase shipping traffic in the Arctic (Laidre et al. 2015, Hovelsrud et al. 2008). One example of this is the year-round shipping through Baffin Bay, planned for the Mary River iron ore project, which will affect the wintering grounds for bearded seals (NAMMCO 2015).
Ship traffic could affect bearded seals and their habitat through their emissions and/or an accidental fuel spill. It could also affect the ecosystem by introducing invasive species or disrupt the seals themselves through the presence of enhanced movement and noise. An increase in noise in the Arctic environment could cause bearded seals to abandon areas of habitat they otherwise might use (Kovacs 2016, Laidre et al. 2015). Increasing ship traffic could also lead to an increased risk in seals being struck by vessels.
Offshore extraction and transportation of oil and gas could affect bearded seals either through direct contact with oil from an accidental spill, or through damage to their habitat and/or prey species.
Contaminants
Contaminants transported to the Arctic from other locations, such as perfluorinated contaminants, organochlorines, and heavy metals like mercury, expose bearded seals to potential risks and environmental hazards.
Mercury in particular is a concern, as it has been found in numerous species across the Arctic in concentrations that exceed guidelines for adverse effects and which may be increasing over time (Dietz et al. 2013). Mercury is a persistent substance that bioaccumulates in living organisms. A recent study in Alaska found both mercury and selenium in bearded seal tissues, with the highest levels found in the liver (Correa et al. 2015).
Studies have detected persistent organochlorine contaminants, including polychlorinated biphenyls (PCBs), Dichlorophenyltrichloroethane (DDT), and chlordane (CHL) related compounds, in the tissues of bearded seals both in the White Sea (Muir et al., 2003) and around Svalbard (Bang et al., 2001).
Bearded seals taken from the Bering and Chukchi Seas have also shown the presence of perfluorinated contaminants (PFCs), such as perfluorooctane sulfonate (PFOS), and related synthetic compounds. However, the levels of these compounds in bearded seals were comparatively low when compared to other species (Quackenbush & Citta, 2008).
Greenland
The Greenland Institute of Natural Resources (GINR) conducts research on bearded seals in Greenland. Ongoing research includes analysis of sound recordings from moorings in West and East Greenland looking at seasonal distribution and movements of bearded seals, especially in relation to seismic activities (NAMMCO 2015; National Progress Report Greenland 2018). There has also been work on satellite tracking, and some diet samples have been collected (NAMMCO 2016).
Greenland participated in a joint survey with Canada of the eastern part of the North Water Polynya in April 2014, to document the importance of this area for overwintering bearded seals and other marine mammals. The most recent aerial survey specifically focusing on bearded seals took place in the spring of 2018 in the North Water Polynya (National Progress Report Greenland 2018).
Norway
In Norway, the Norwegian Polar Institute is responsible for conducting research on bearded seals, with a significant focus on research activities carried out in Svalbard. There has been some passive acoustic monitoring to record bearded seal and other marine mammal vocalisations. This leads to information about bearded seal distribution throughout the year. Bearded seals have also been tagged in Svalbard and this work and analysis is ongoing (Lydersen et al. 2014).
First working group meeting currently planned for 2022.
References
Andersen, M., Hjelset, A. M., Gjertz, I., Lydersen, C. and Gulliksen, B. (1999). Growth, age at sexual maturity and condition in bearded seals (Erignathus barbatus) from Svalbard, Norway. Polar Biology, 21, 179–185. https://doi.org/10.1007/s003000050350
Antonelis, G.A., Melin, S.R. and Bukhtiyarov, Y.A. (1994). Early Spring Feeding Habits of Bearded Seals (Erignathus barbatus) in the Central Bering Sea, 1981. Arctic, 47(1), 74–79. https://doi.org/10.14430/arctic1274
Bang, K., Jenssen, B.M., Lydersen, C. and Skaare, J.U. (2001). Organochlorine burdens in blood of ringed and bearded seals from north-western Svalbard. Chemosphere, 44(2), 193–203. https://doi.org/10.1016/S0045-6535(00)00197-1
Cameron, M. F., Bengtson, J.L., Boveng, P.L., Jansen, J.K., Kelly, B.P., Dahle, S.P., Logerwell, E.A. Overland, J.E., Sabine, C.L., Waring, G.T. and Wilder, J.M. (2010). Status review of the bearded seal (Erignathus barbatus ). U.S. Dep. Commer., NOAA Tech. Memo. NMFS-AFSC-211, 246 p. Available at https://www.fisheries.noaa.gov/resource/document/status-review-bearded-seal-erignathus-barbatus
Charrier, I., Mathevo, N. and Aubin, T. (2013). Bearded seal males perceive geographic variation in their trills. Behavioral Ecology and Sociobiology, 67, 1679–1689. https://doi.org/10.1007/s00265-013-1578-6
Cleator, H.J. (1996). The status of the bearded seal, Erignathus barbatus, in Canada. Canadian Field Naturalist, 110, 501–510.
Correa, L., Castellini, J. M., Quackenbush, L.T. and O’Hara, T.M. (2015). Mercury and Selenium concentrations in skeletal muscle, liver, and regions of the heart and kidney in bearded seals from Alaska, USA. Environmental Toxicology and Chemistry, 34(10), 2403–2408. https://doi.org/10.1002/etc.3079
Davis, C.S., Stirling, I., Strobeck, C. and Coltman, D.W. (2008). Population structure of ice‐breeding seals. Molecular Biology, 17(13), 3078‐3094. https://doi.org/10.1111/j.1365-294X.2008.03819.x
Department of Fisheries and Oceans Canada (DFO). (2016). 2011-2015 Integrated Fisheries Management Plan for Atlantic Seals. http://www.dfo-mpo.gc.ca/fm-gp/seal-phoque/reports-rapports/mgtplan-planges20112015/mgtplan-planges20112015-eng.htm
Dietz,R., Sonne, C., Basu, N., Braune, B., … and Aars, J. (2013). What are the toxicological effects of mercury in Arctic biota? Science of the Total Environment, 443, 775–790. https://doi.org/10.1016/j.scitotenv.2012.11.046
Dixon, B. R., Parrington, L.J., Parenteau, M., Leclair, D., Santín, M. and Fayer, R. (2008). Giardia duodenalis and Cryptosporidium spp. in the intestinal contents of ringed seals (Phoca hispida) and bearded seals (Erignathus barbatus) in Nunavik, Quebec, Canada. Journal of Parasitology, 94(5), 1161‐1163. https://doi.org/10.1645/GE-1485.1
EUR-Lex. (2016). Access to European Union Law. Available at http://eur-lex.europa.eu/legal-content/EN/LSU/?uri=CELEX:32009R1007&qid=1455217489306
Finlay, K. and Evans, C. R. (1983). Summer Diet of the Bearded Seal (Erignathus barbatus) in the Canadian High Arctic. Arctic, 36(1), 82–89. https://doi.org/10.14430/arctic2246
Forbes, L. B. (2000). The occurrence and ecology of Trichinella in marine mammals. Veterinary Parasitology, 93(3–4), 321‐334. https://doi.org/10.1016/S0304-4017(00)00349-6
Greenland Institute of Natural Resources (GINR). (2016). Bearded seal. Available at http://www.natur.gl/en/birds-and-mammals/marine-mammals/bearded-seal/
Gjertz, I., Kovacs, K. M., Lydersen, C. and Wiig, Ø. (2000). Movements and diving of bearded seal (Erignathus barbatus) mothers and pups during lactation and post-weaning. Polar Biology, 23, 559–566. https://doi.org/10.1007/s003000000121
Hammill, M., Kovacs, K.M. and Lydersen, C. (1994). Local movements by nursing bearded seal (Erignathus barbatus) pups in Kongsfjorden, Svalbard. Polar Biology, 14, 569–570. https://doi.org/10.1007/BF00238227
Hauksson, E., and Bogason, V. (1995). Occurrences of bearded seals (Erignathus barbatus Erxleben, 1777) and ringed seal (Phoca hispida Schreber, 1775) in Icelandic waters, in the period 1990‐1994, with notes on their food. Council Meeting of the International Council for the Exploration of the Sea CM 1995/N:15.
Hindell, M.A., Lydersen, C., Hop, H. and Kovacs, K.M. (2012). Pre-Partum Diet of Adult Female Bearded Seals in Years of Contrasting Ice Conditions. PLoS ONE, 7(5), e38307. https://doi.org/10.1371/journal.pone.0038307
Hovelsrud, G.K., McKenna, M. and Huntington, H.P. (2008). Marine mammal harvests and other interactions with humans. Ecological Applications, 18(2), S135–S147.https://doi.org/10.1890/06-0843.1
Jones, J.M., Thayre, B.J., Roth, E. H., Mahoney, M., Sia, I., Merculief, K., Jackson, C., Zeller, C., Clare, M., Bacon, A., Weaver, S., Gentes, Z., Small, R.J., Stirling, I., Wiggins, S.M., Hildebrand,J. A. and Giguère, N. (2014). Ringed, Bearded, and Ribbon Seal Vocalizations North of Barrow, Alaska: Seasonal Presence and Relationship with Sea Ice. Arctic, 67(2), 203–222. https://doi.org/10.14430/arctic4388
Kovacs, K.M. (2016). Erignathus barbatus. The IUCN Red List of Threatened Species 2016: e.T8010A45225428. http://dx.doi.org/10.2305/IUCN.UK.2016-1.RLTS.T8010A45225428.en.
Kovacs, K.M., Moore, S., Overland, J.E. and Lydersen, C. (2011). Impacts of changing sea-ice conditions on Arctic marine mammals. Marine Biodiversity, 41, 181–194. https://doi.org/10.1007/s12526-010-0061-0
Kovacs, K.M., Lydersen, C. and Gjertz, I. (1996). Birth-site characteristics and prenatal molting in bearded seals (Erignathus barbatus). Journal of Mammalogy, 77(4), 1085–1091. https://doi.org/10.2307/1382789
Laidre, K.L., Stern, H., Kovacs, K.M., Lowry, L., … and Ugarte, F. (2015). Arctic marine mammal population status, sea ice habitat loss, and conservation recommendations for the 21st century. Conservation Biology, 29(3), 724–737. https://doi.org/10.1111/cobi.12474
Lydersen, C. and Kovacs, K. M. (1999). Behaviour and energetics of ice-breeding, North Atlantic phocid seals during the lactation period. Marine Ecology Progress Series, 187, 265–281. https://doi.org/10.3354/meps187265
Lydersen, C., Assmy, P., Falk-Petersen, S., Kohler J., … and Zajaczkowski, M. (2014). The importance of tidewater glaciers for marine mammals and seabirds in Svalbard, Norway. Journal of Marine Systems, 129, 452–471. https://doi.org/10.1016/j.jmarsys.2013.09.006
Lydersen, C., Kovacs, K.M. , Hammill, M.O. and Gjertz, I. (1996). Energy intake and utilization by nursing bearded seal (Erignathus barbatus) pups from Svalbard, Norway. Journal of Comparative Physiology B, 166, 405–411. https://doi.org/10.1007/BF02337884
Lydersen, C., Hammill, M.O. and Kovacs, K.M. (1994). Diving activity in nursing bearded seal (Erignathus barbatus) pups. Canadian Journal of Zoology, 72(1), 96–103. https://doi.org/10.1139/z94-013
MacIntyre, K.Q., Stafford, K.M., Berchok, C. L. and Boveng, P.L. (2013). Year-round acoustic detection of bearded seals (Erignathus barbatus) in the Beaufort Sea relative to changing environmental conditions, 2008–2010. Polar Biology, 36, 1161–1173. https://doi.org/10.1007/s00300-013-1337-1
Marshall, C.D. (2016). Morphology of the bearded seal (Erignathus barbatus) muscular– vibrissal complex: a functional model for phocid subambient pressure generation. The Anatomical Record, 299(8), 1043–1053. https://doi.org/10.1002/ar.23377
Mizuguchi, D., Tsunokawa, M., Kawamoto, M. and Kohshima, S. (2016). Sequential calls and associated behavior in captive bearded seals (Erignathus barbatus ). Journal of the Acoustical Society of America, 140(4), 3238–3238. https://doi.org/10.1121/1.4970242
Muir, D., Savinova, T., Svinov, V., Alexeeva, L., Potelov, V. and Svetochev, V. (2003). Bioaccumulation of PCBs and chlorinated pesticides in seals, fishes and invertebrates from the White Sea, Russia. Science of the Total Environment, 306(13), 111–131. https://doi.org/10.1016/S0048-9697(02)00488-6
North Atlantic Marine Mammal Commission (NAMMCO). (2016). Report of the 23rd of the Scientific Committee Meeting, Nuuk, Greenland, 4-7 November. 291 pp. https://nammco.no/topics/scientific-committee-reports/
North Atlantic Marine Mammal Commission (NAMMCO). (2015). Annual report 2015. NAMMCO, Tromsø, Norway. 374 p. https://nammco.no/topics/annual-reports/
North Atlantic Marine Mammal Commission (NAMMCO). (2014). Annual report 2014. NAMMCO, Tromsø, Norway. 246 p. https://nammco.no/topics/annual-reports/
North Atlantic Marine Mammal Commission (NAMMCO). (2009). Annual Report 2007‐2008. NAMMCO, Tromsø, Norway. 399 p. https://nammco.no/topics/annual-reports/
Priest, H. and Usher, P.J. (2004). The Nunavut Wildlife Harvest Study. Nunavut Wildlife Management Board. 816 p. Available at https://www.nwmb.com/inu/publications/harvest-study/1824-156-nwhs-report-2004-156-0003/file
Quackenbush, L.T. and Citta, J.J. (2008). Perfluorinated contaminants in ringed, bearded, spotted, and ribbon seals from the Alaskan Bering and Chukchi Seas. Marine Pollution Bulletin, 56(10), 1802–1814. https://doi.org/10.1016/j.marpolbul.2008.06.005
Richard, P. (2001). Marine mammals of Nunavut. Qikiqtani School Operations, Iqaluit NU. pp.98.
Risch, D., Clarke, C.W., Cockeron, P.J., Elepfandt, A., Kovacs, K.M., Lydersen, C., Stirling, I. and Van Parijs, S.M. (2007). Vocalizations of male bearded seals, Erignathus barbatus: classification and geographical variation. Animal Behavior, 73(5), 747–762. https://doi.org/10.1016/j.anbehav.2006.06.012
Sardi, K. A. and Merigo, C. (2006). Erignathus barbatus (Bearded seal) Vagrant in Massachusetts. Northeastern Naturalist, 13(1), 39–42. https://doi.org/10.1656/1092-6194(2006)13[39:EBBSVI]2.0.CO;2
Stewart, D.B. and Lockhart, W.L. (2005). An overview of the Hudson Bay Marine Ecosystem. Canadian Technical Report of Fisheries and Aquatic Sciences, 2586, vi + 487 p. https://waves-vagues.dfo-mpo.gc.ca/Library/314704covermaterial.pdf
Van Parijs, S.M. and Clark, C.W. (2006). Long-term mating tactics in an aquatic-mating pinniped, the bearded seal, Erignathus barbatus. Animal Behaviour, 72(6), 1269–1277. https://doi.org/10.1016/j.anbehav.2006.03.026
Van Parijs, S.M., Lydersen, C. and Kovacs, K.M. (2004). The effects of ice cover on the behavioural patterns of aquatic mating male bearded seals. Animal Behavior, 68(1), 89–96. https://doi.org/10.1016/j.anbehav.2003.09.013
Norwegian Polar Institute – Bearded seal
Greenland Institute of Natural Resources – Bearded seal
Canadian Dept of Fisheries and Oceans – Bearded seal
US National Oceanic and Atmospheric Administration (NOAA) Fisheries -Bearded seal
Alaskan Department of Fish and Game – Bearded seal
Seal Conservation Society – Bearded seal
Ocean Conservation Research – Bearded seal (including audio recordings of vocalisations)
Discovery of Sound in the Sea – Bearded seal vocalisations
VIDEOS: